Consider this; a future where your favorite steak is not only tender and flavorful but also produced with minimal environmental impact, free from antibiotics, and cruelty-free. Now, what if it’s not the future but today’s reality brought about by food innovation efforts over the years? This is the notion of cultivated meat.
As we stand on the brink of a food revolution, the scale-up and bioprocessing of cultivated meat are key to transforming this vision into an everyday staple. In tandem, ensuring rigorous food safety standards throughout the process as cultivated meat moves from the lab to our plates is key to earning consumer acceptance and confidence.
What is Cultivated Meat?
Cultivated meat, also referred to as cultured meat, is authentic animal meat (including seafood and organ meats) produced by cultivating animal cells directly. This innovative method eliminates the need to raise and farm animals for food. Cultivated meat consists of the same cell types arranged in similar structures to animal tissues, replicating the sensory and nutritional profiles of conventional meat.
The first cultivated meat burger was unveiled by Dutch scientist Mark Post on live television in 2013. By 2015, the first four companies dedicated to cultivated meat were established. As of late 2022, the industry has expanded to over 150 companies across six continents, all aiming to produce cultivated meat products.
Fundamental Processes Involved in Cultivated Meat Production
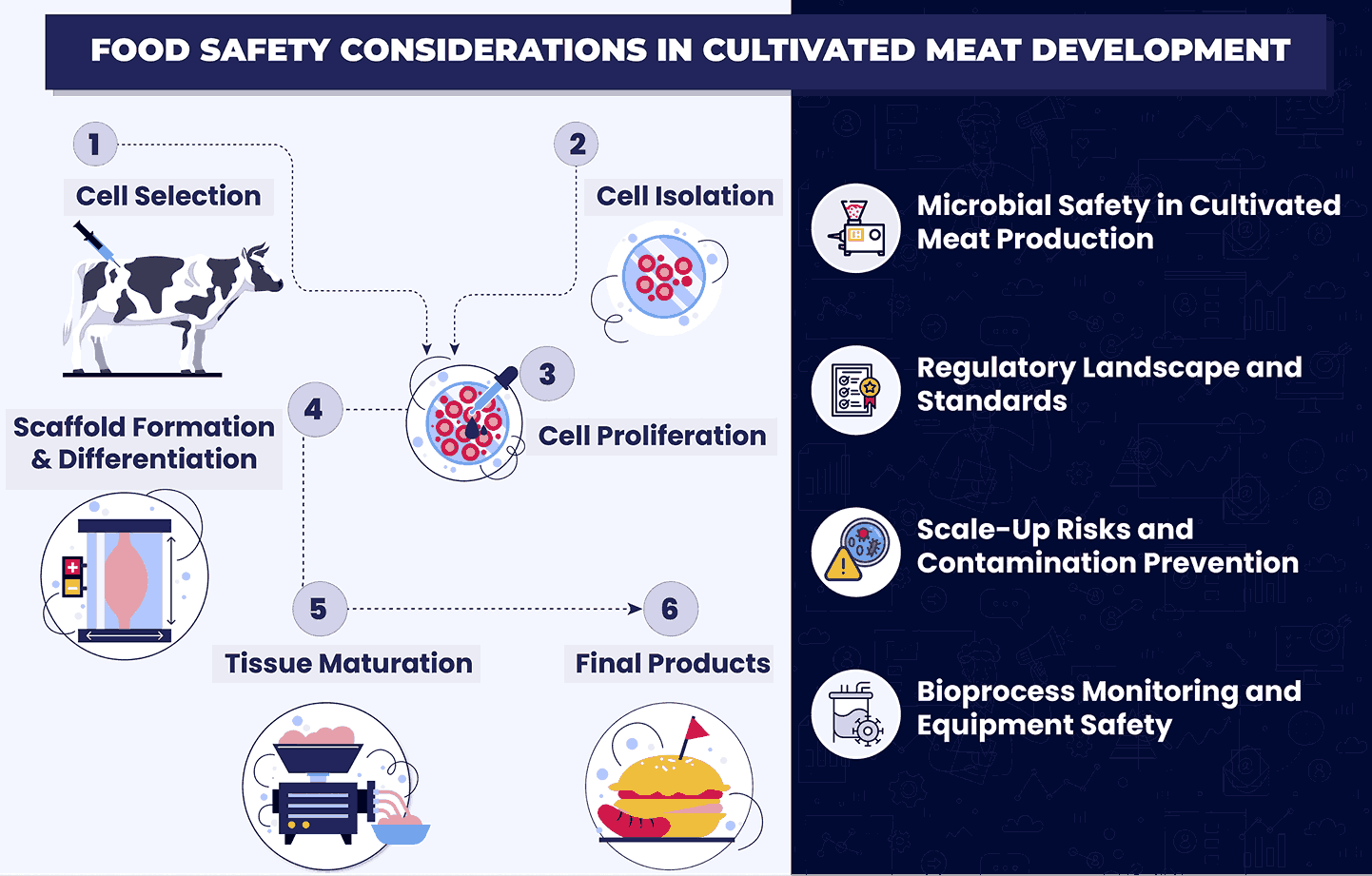
The process of cultivated meat scale-up and bioprocessing can be broken down into the following several stages:
1. Cell Selection and Isolation
The journey of cultivated meat begins with the careful selection and isolation of animal cells. These cells can be stem cells or muscle cells, typically obtained through a small biopsy from a living animal. The choice of cells is crucial, as they must have the potential to proliferate and differentiate into various tissues such as muscle, fat, and connective tissue.
2. Cell Proliferation
Once isolated, the cells are placed in a controlled environment known as a bioreactor. Here, they are immersed in a nutrient-rich culture medium containing essential nutrients, growth factors, and hormones. This medium recreates the environment that cells would naturally encounter within an animal’s body, stimulating rapid cell multiplication. This step transforms a small initial sample into a large number of cells, providing the raw material needed for meat production.
3. Scaffold Formation
To develop the texture and structure of meat, the proliferated cells need to grow in three dimensions. This is achieved by combining the cells with a scaffold, which is typically made of edible, biodegradable materials, carrying nutrients to support further cell differentiation. The scaffold acts as a framework for the cells to attach to and grow, mimicking the natural formation of muscle tissue.
4. Differentiation
The cells within the bioreactor are then induced to differentiate into specific types, such as muscle or fat cells, by altering the conditions of the culture medium. Proper differentiation ensures that the cultivated meat closely resembles conventional meat in terms of its composition and culinary properties.
5. Tissue Maturation
After differentiation, the cells are allowed to mature into fully developed tissue. This maturation process can involve mechanical or electrical stimulation to enhance the development of muscle fibers, closely replicating the natural growth of animal muscle tissue.
6. Harvesting
Once the cells have matured into the desired tissue, the cultivated meat is ready for harvesting. This involves extracting the tissue from the bioreactor and processing it into final meat products, such as burgers, sausages, or fillets.
Food Safety Considerations in the Development of Cultivated Meat
The rise of cultivated meat represents a paradigm shift in food production, promising to meet growing protein demands while mitigating the environmental impacts of conventional livestock farming. However, scaling up the production of cultivated meat presents unique challenges, particularly in maintaining food safety. Let’s look into the different aspects of food safety of cultivated meat:
⇒ Microbial Safety in Cultivated Meat Production
Microbial contamination is a primary concern in food production, and cultivated meat is no exception. However, cultivated meat production has inherent advantages that can reduce contamination risks compared to traditional meat processing.
The initial product in cultivated meat consists of pathogen-free cells derived from aseptic processes, considered cleaner than meat from slaughterhouses. This reduces the risk of introducing contaminants such as animal fecal microorganisms and zoonotic diseases. Despite these advantages, the cultivation process itself is prone to contamination due to the nutrient-rich media used for cell growth, which can harbor bacteria, molds, and yeasts.
Recommended Actions: Companies in the cultivated meat industry must implement stringent contamination prevention strategies, including sterile culture media, high-quality raw materials, and extensive testing to discard contaminated materials.
⇒ Regulatory Landscape and Standards
The regulatory landscape for cultivated meat is evolving, with new standards and regulations being developed to ensure its safety globally. Regulatory authorities and food safety organizations play a main role in shaping these standards.
Recommended Actions: A significant aspect of maintaining food safety is the implementation of Hazard Analysis and Critical Control Points (HACCP) plans. These plans involve identifying potential hazards, developing controls, and implementing a comprehensive food safety plan. Many companies in the cultivated meat sector have adopted HACCP plans, some of which have been certified by third-party organizations such as ISO. Good Manufacturing Practices (GMP) certifications can be pursued to further ensure product safety.
⇒ Scale-Up Risks and Contamination Prevention
Scaling up cultivated meat production from lab-scale to commercial scale introduces several challenges, particularly in maintaining contamination-free environments. Managing contamination at scale is complex, requiring regular testing of cell banks and rigorous contamination checks throughout the bioprocessing stages. Companies have identified process contamination, final product contamination, and raw material contamination as primary sources of microbial risk.
Recommended Actions: One of the critical strategies for preventing contamination is the use of automated procedures and well-controlled biopharmaceutical procedures. The use of sterilized bioreactors and automated methods can significantly reduce contamination risks. Regular monitoring and quality control measures, such as microbial testing and contamination checks, are essential for ensuring the final product’s safety.
⇒ Post-Production Quality Control
Post-production quality control (QC) is pivotal for ensuring the safety and quality of cultivated meat products. Companies conduct various tests, including microbial testing, lipid composition analysis, and spent media analysis, to validate the nutritional value and detect any contaminants. These tests help in assessing the efficiency of the media and the overall health of the cell cultures.
Recommended Actions: Microbial testing and contamination checks are the most frequently conducted QC analyses, emphasizing the industry’s focus on minimizing contamination risks. Shelf-life determination and antibiotic testing can also be performed to ensure product safety and compliance with regulatory standards.
⇒ Bioprocess Monitoring and Equipment Safety
Effective bioprocess monitoring is vital for maintaining food safety during cultivated meat production. This involves continuous monitoring of cell culture conditions, media quality, and bioreactor performance. The choice of equipment also plays a significant role in ensuring food safety. While the industry initially borrowed technologies from the pharmaceutical sector, there is now a shift towards developing fit-for-purpose equipment for cultivated meat production.
Recommended Actions: Companies are required to employ advanced diagnostic and testing approaches, such as immunostaining diagnostics and cell viability monitoring, to detect and mitigate potential issues early in the process. Using food-grade rather than pharmaceutical-grade materials can reduce production costs while maintaining safety. Companies must balance the need for high-quality equipment with the economic feasibility of scaling up production.
Smart Food Safe as a One-Stop Digital Solution for Enhancing Food Safety in the Cultivated Meat Industry
Innovative food production and processing demands innovative means of ensuring food safety. Smart Food Safe strives to be an enabler for innovative food sectors such as the cultivated meat industry by providing a suite of digital modules embedded with diverse functions critical to food safety in cultivated meat scale-up and bioprocessing operations. Here’s how Smart Food Safe assists:
Process Tracking & Traceability
Batch Tracking: Smart Record further becomes instrumental to extensively track and managing batch records, which is crucial for identifying and isolating any contamination sources quickly.
End-to-End Traceability: Provides detailed traceability from the raw materials (cells) to the final product, ensuring every step in the bioprocessing chain is documented and verifiable.
Continuous Monitoring & Compliance
Regulatory Compliance: Smart Food Safe’s Smart Docs equips cultivated meat producers to adhere to cultivated meat-specific food safety standards and regulations such as FDA, EFSA, and other local regulatory bodies.
Real-time Monitoring: Constant monitoring of critical control points (CCPs) and key parameters in bioprocessing promotes the ability to detect and address potential safety issues immediately.
Non-conformance Management: Smart CAPA competently handles non-conformance incidents with root cause analysis and corrective action tracking to prevent recurrence.
Data Integration & Environmental Hygiene
Smart Integration: Smart Food Safe enables intermodular as well as external integration with hardware and software devices for uninterrupted data integration from various sources like bioreactors, laboratories, and production lines, allowing for comprehensive analysis and decision-making.
Pathogen Detection: Smart EMP incorporates features to depict potential safety hazards in the production facilities and process deviations before they escalate, reducing risks associated with cultivated meat production.
Sanitation Schedules: Smart EMP can be employed as part of sanitation schedules in order to maintain compliance, ushering in a clean and safe production environment.
Training and Documentation
Employee Training: Smart Training provides employee training functionalities to ensure all personnel are knowledgeable about safety protocols and best practices in cultivated meat production.
Documentation Management: Maintains comprehensive documentation of all processes, inspections, and safety protocols, facilitating audits and reviews.

