
Food Safety in March 2026: Outbreaks, Recalls & What Industry Must Do Next
March 2026 was defined by a convergence of outbreaks, recalls, and regulatory signals that together highlight evolving risks and expectations across the food industry. From an ongoing E. coli outbreak to 97 Salmonella cases and a rise in allergen recalls, the month reinforced how existing systems are being tested.

There's rarely a quiet month in food safety, and March 2026 was no exception. Two major outbreak investigations unfolded in parallel. A supplement ingredient linked to a toxic plant expanded into a growing list of affected products. Allergen mislabelling continued to appear across categories. At the same time, regulators introduced new digital tools and opened key policy discussions, signalling a broader shift toward more transparent, data-driven compliance.
What follows is a clear, fact-based breakdown of what happened in March supported by verified data and source references, so you can understand not just the events, but what they mean for food safety systems moving forward.
Ongoing Outbreak
1. E. coli O157:H7 Linked to Raw Cheddar Cheese

Nine illnesses, three states, one unresolved recall standoff. More than half the victims are children under five.
The FDA and CDC opened a multistate E. coli O157:H7 investigation in March that remained active as of month's end. The investigation traces back to raw cheddar cheese sold at major retailers.
The CDC issued a formal public warning on March 16, 2026. Since the last update on March 15, two additional illnesses were reported, bringing the total to nine.
Of eight people interviewed, all reported consuming raw dairy, and seven pointed to the same source. Whole genome sequencing confirmed a single common origin. Illness onset dates stretch from September 2025 to February 2026, meaning exposure was months underway before any public alert.
Watch Point
One case resulted in hemolytic uremic syndrome (HUS), a life-threatening kidney complication most common in young children. When voluntary recall authority is refused during an active investigation, FDA's enforcement options are limited and contaminated product stays available. This case puts that gap in sharp focus.
Practical Takeaway
Strengthen traceback depth across raw dairy and raw ingredient supply chains. Know your hold-and-release process before an outbreak forces the question.
Closed Outbreak + Active Investigation
2. Moringa Powder Outbreak Closes at 97 Cases in 32 States

One investigation closed on March 17. By that point, it had reached 97 confirmed illnesses with a hospitalisation rate higher than the Salmonella average. A second, drug-resistant outbreak is still open.
On March 17, 2026, FDA and CDC formally closed a Salmonella Typhimurium and Newport outbreak tied to recalled moringa leaf powder supplements. Traceback connected two separately branded products to a single common ingredient manufacturer. Lab results found the outbreak strain in two raw moringa ingredient samples.
Wisconsin led in case count at 12, followed by New York (8) and Minnesota (7). The 29% hospitalisation rate sits well above the typical 20% for Salmonella outbreaks. Products had best-by dates extending to 2028, meaning recalled items may still sit in consumers' homes.
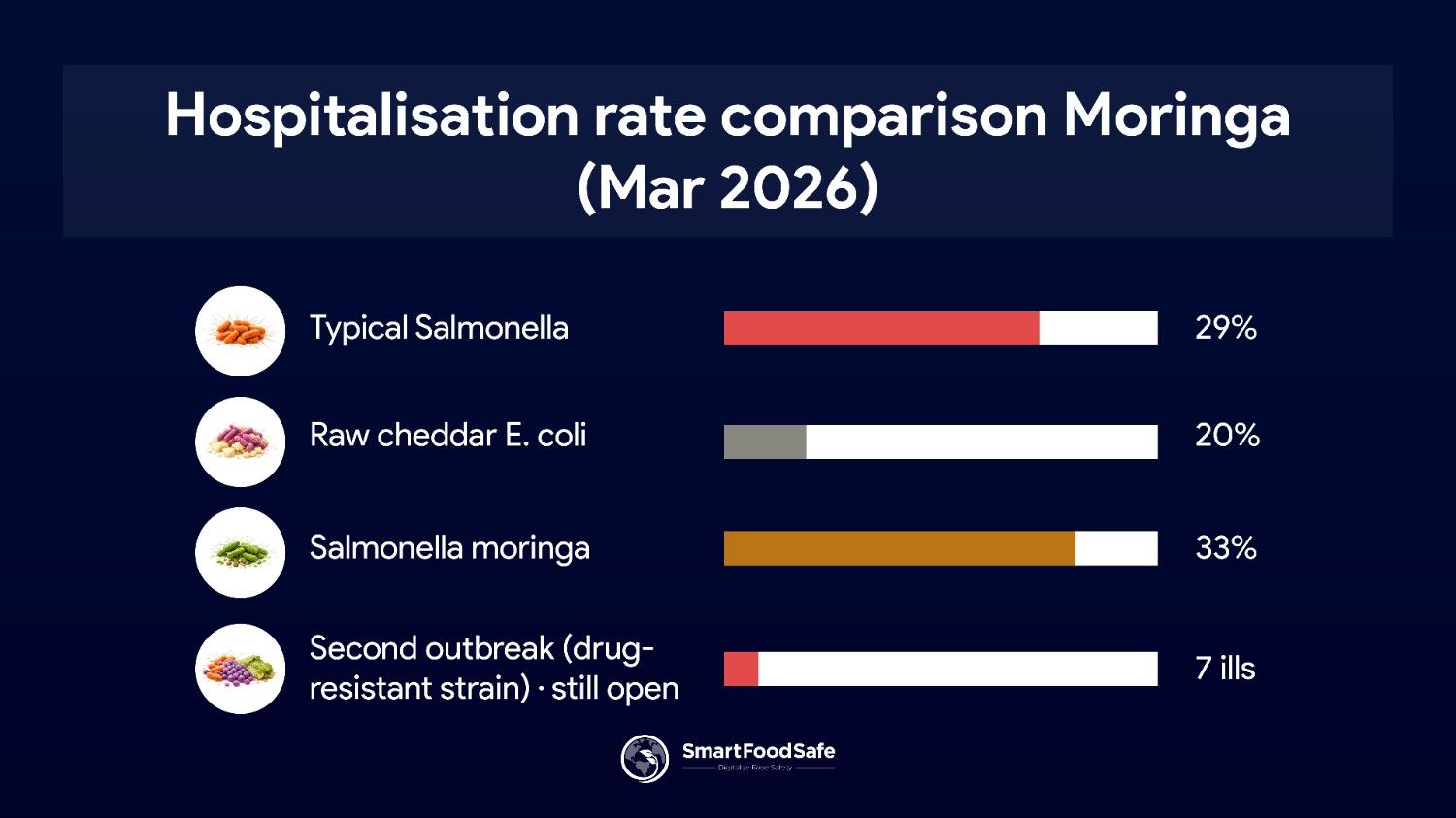
Second Outbreak Still Active
A separate investigation announced February 13 links seven illnesses and three hospitalisations across seven states to moringa capsules under a different label. The strain involved is extensively drug-resistant, meaning it is resistant to all first-line and alternative antibiotics. As of March 24, the investigation was still open.
Practical Takeaway
Botanical supplements with long shelf lives extend the hazard window significantly. Incoming-material pathogen testing and shared-manufacturer transparency are the only controls that catch this kind of failure before it reaches consumers.
Allergen Recalls
3. Allergen Contamination Drives Nearly Half of All US Food Recalls
March's recall list stretched across seven product categories. The root cause in nearly every case: a process failure that was preventable.
Allergen contamination accounts for 45.2% of all US food recall cases, more than double the next most common cause at 22.6% for bacterial contamination. Class I recalls, those with the highest potential for serious harm, grew 36.4% between 2021 and 2025.
Source: Trace One analysis of 2021–2025 FDA data
March FDA listings covered undeclared allergen recalls across these product categories:
Almonds in cream cheese spread
Milk in potato chips
Wheat and soy in honey BBQ sauce
Hazelnut in chocolate candy
Egg, hazelnut and soy in dietary supplement
Peanuts in frozen cookie dough
Soy lecithin in dried beef snack (FSIS alert)
One case illustrates how long these failures can go undetected: a honey-based BBQ sauce was mislabelled for over seven months (July 2025 to February 2026) before the error surfaced. No illnesses were reported, but national distribution was complete before the recall was issued.
In a separate case, frozen cookie dough containing peanuts was packaged into bags labelled as a nut-free variety and distributed across three states before the error was caught.
Pattern Note
Both cases trace back to the same root causes found across most allergen recalls: labels that no longer match the formulation, and no pre-release check to catch it. These are process gaps, and they are entirely preventable.
Practical Takeaway
Verify final labels against the actual formulation on every production run. Confirm allergen segregation at both the ingredient and equipment level. A pre-release verification step before any product leaves the facility prevents a seven-month mislabelling window.
Supplement Safety Alert
4. 29 Supplement Products Found to Contain Yellow Oleander, a Toxic Plant

Sold as a weight-loss root extract. Found to contain a poisonous plant. Some products remain available online despite FDA warnings.
FDA's updated safety alert, expanded again in March 2026, now covers 29 supplement products labelled as tejocote root or Brazil seed that were found on testing to contain yellow oleander (Thevetia peruviana), a toxic plant native to Mexico and Central America. Consuming yellow oleander can cause arrhythmia, convulsions, vomiting, and potentially fatal cardiovascular effects.
On March 11, FDA's Human Foods Program announced an additional product added to the list, sold via eBay. Products continued to appear on Amazon, eBay, and Etsy throughout the month. Several firms have not responded to FDA contact, and some have refused voluntary recalls.
This Is Deliberate Substitution
You cannot distinguish yellow oleander from tejocote root by appearance. Identity testing on incoming material is the only reliable control here. Supplier certification alone is insufficient when the substitution occurs at the ingredient level before the product reaches a formulator.
Practical Takeaway
Check FDA's full product list if you stock, formulate with, or retail any tejocote root or Brazil seed products. If you have sold any flagged lots, consumer notification and lot tracing should begin immediately.
FSIS Public Health Alerts
5. Five FSIS Public Health Alerts in March: Metal, Misbranding and Uninspected Product
Public health alerts often move faster than formal recalls. March had five in under four weeks, covering four distinct hazard types.
March 1
Dried beef snack products flagged for misbranding and undeclared soy lecithin allergen, establishment EST. 2625.
March 9
Frozen ready-to-eat meal recall scope expanded: all units with affected lot numbers added, regardless of best-by date.
March 13
Frozen turkey stuffed pastry products flagged for misbranding: boxes labelled turkey, pesto and Swiss found to contain undeclared ham.
March 23
16 oz vacuum-packed grassfed ground beef alerted for possible metal foreign material. Product was no longer in trade, but consumer alert issued for product already in homes.
March 25
Raw beef and pork products flagged for being produced without federal inspection and bearing a falsified USDA mark of inspection. A simultaneous compliance and food safety violation.

Practical Takeaway
Public health alerts signal serious risk before the formal recall process is complete. The correct response sequence: stop sale, verify lots, notify downstream distribution partners, and escalate internally, do not wait for an official recall notice.
Regulatory Moves
6. Three March Regulatory Actions That Signal Where Compliance Is Heading
No dramatic rule changes this month. But three policy moves point clearly at the direction FDA and FSIS expect industry to move in next.
Gluten labelling comment deadline extended to April 22
FDA extended the comment period for its Labeling and Preventing Cross-Contact of Gluten for Packaged Foods request for information from March 23 to April 22, 2026, under docket FDA-2023-P-3942. The RFI focuses on rye, barley, and oats on shared equipment, and had already received over 3,300 comments before the extension. Federal Register National Law Review
Public meeting on dietary supplement ingredient scope, March 27
FDA's Office of Dietary Supplement Programs held a hybrid public meeting to explore how dietary ingredient categories under DSHEA apply to novel production methods, proteins, enzymes, and microbials. Comments remain open until April 27, 2026 under docket FDA-2026-N-2047. Industry observers have called this a potential reopening of a definitional question that has been largely dormant for 32 years. FDA NutraIngredients
FSIS Directive 7120.1 Revision 60 and new developer tools
FSIS issued Revision 60 of Directive 7120.1, updating the list of safe and suitable ingredients for meat, poultry, and egg products, and maintaining it as a live digital database. The accompanying developer resources page provides API access to recall data and inspection information, allowing real-time ingredient verification rather than manual lookups against static documents. FSIS Directive FSIS Dev Tools
What These Three Moves Share
Each one pushes toward the same expectation: faster verification, better documentation, and compliance data that lives inside operational workflows rather than in binders checked once a year. When a regulator publishes an API, it is a signal that manual-only compliance is no longer the expected standard.
What March 2026 tells us about
where food safety is heading
where food safety is heading
What March 2026 tells us about where food safety is heading
The System Is Working. Now Make It Work for You.
Here is something worth sitting with: the Salmonella moringa outbreak was closed in under six months, across 32 states, with zero deaths. Whole genome sequencing linked all nine E. coli cases to a single source before a second wave could form. A toxic plant substitution affecting 29 products was caught through lab testing, not a hospitalisation cluster. These outcomes are not accidental. They reflect a food safety infrastructure that has genuinely improved at finding problems faster and containing them before they compound.
The tools for early detection exist and are getting better. Regulators are publishing real-time APIs, tightening traceback expectations, and opening policy consultations that invite industry input before rules are written. The direction is transparent and the runway is visible. For businesses willing to move with it rather than react to it, that is an advantage.
Prevention compounds just like risk does. Every allergen control tightened today, every supplier verified this quarter, every label checked before a product ships is a future recall that never happens, a consumer who never gets hurt, and a brand that never has to rebuild trust from scratch. The cost of prevention is always smaller than people expect. The cost of recovery is always larger.
Primary sources
FDA: E. coli Outbreak (March 2026) · CDC: E. coli Update · FDA: Salmonella Moringa Outbreak · FDA: Yellow Oleander Alert · FSIS: Recalls and Alerts · Federal Register: Gluten RFI Extension · FSIS Directive 7120.1 Rev. 60 · Trace One: 2025 Recall Analysis
All links verified as of March 30, 2026. For informational purposes only.
FDA: E. coli Outbreak (March 2026) · CDC: E. coli Update · FDA: Salmonella Moringa Outbreak · FDA: Yellow Oleander Alert · FSIS: Recalls and Alerts · Federal Register: Gluten RFI Extension · FSIS Directive 7120.1 Rev. 60 · Trace One: 2025 Recall Analysis
All links verified as of March 30, 2026. For informational purposes only.


