If you are familiar with the basic principle of toxicology, you must have heard the saying by Swiss physician and chemist Paracelsus, “The dose makes the poison.” The same precept can be applied to the food additives we consume, meaning they can potentially become a hazard beyond a certain level. This is why it is important to examine carefully the development and usage of food additives.
This being said, the United States and the European Union (EU) take distinct approaches to risk management with the primary food safety regulatory bodies, the U.S. Food and Drug Administration (FDA) and the European Food Safety Authority (EFSA), evaluating and interpreting scientific studies on various food additives or ingredients from differing perspectives. These differences can significantly impact how food manufacturers formulate and market food additives, especially multi-nationally in an increasingly globalized market.
Basics of Food Additive
Food additives are substances added to food products to enhance flavor, texture, appearance, or shelf life. These additives can be natural or synthetic and are used in a variety of ways, such as preservatives to prevent spoilage, colorants to make foods visually appealing, and flavor enhancers to improve taste. Additives are classified into several categories, including preservatives, colorants, flavorings, and stabilizers.
Each additive is subject to rigorous safety evaluations by regulatory agencies to ensure they are safe for consumption at specified levels. The use of food additives is regulated by governmental bodies, such as the FDA in the United States or EFSA in Europe, which set standards for acceptable levels and required labeling.
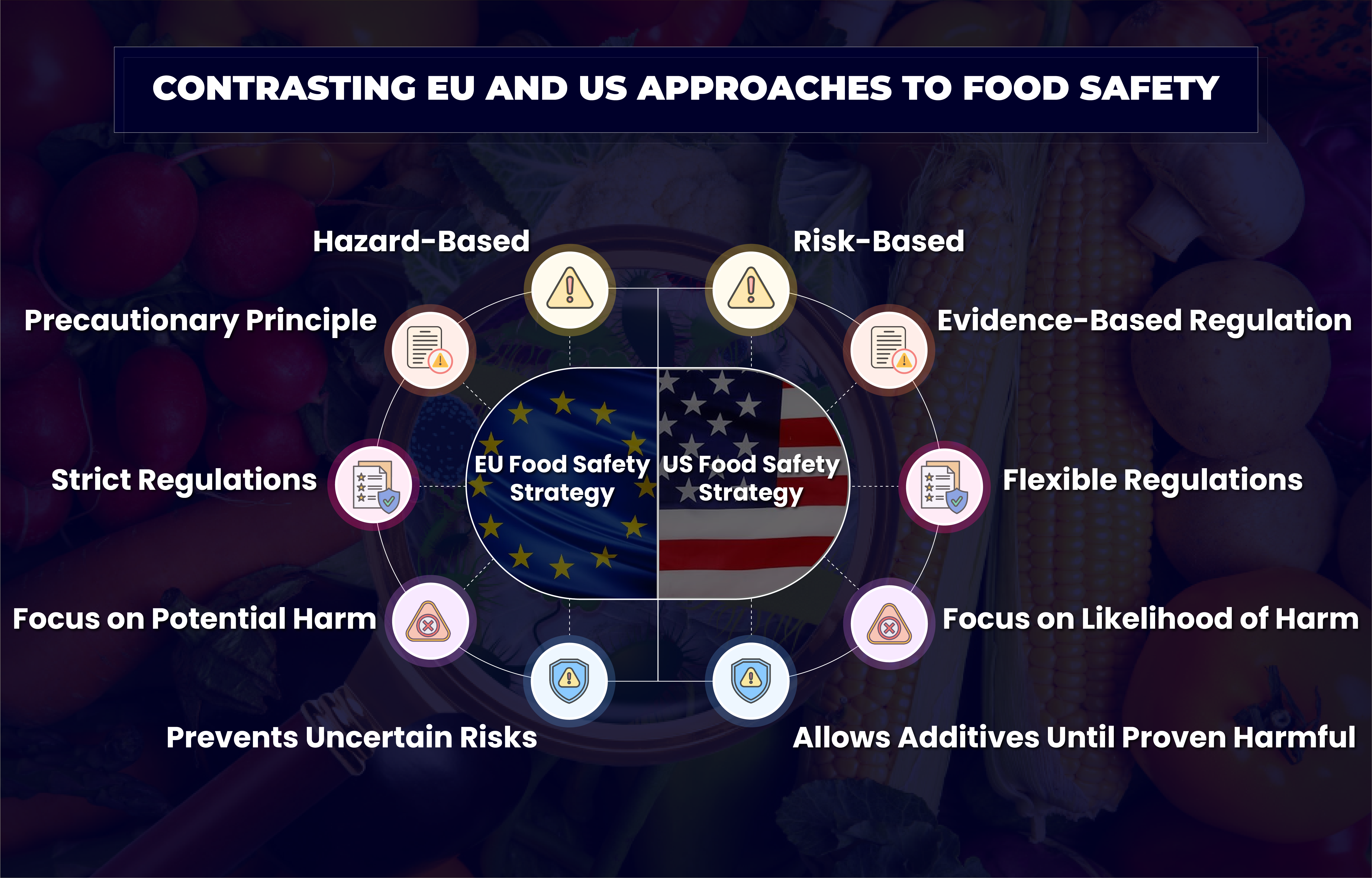
Hazard vs. Risk: Core Difference in FDA & EFSA Food Safety Strategies
Steve Havlik, a former senior director at PepsiCo rightly explains, “The U.S. tends to be more hands-off until there’s proof that something is harmful, whereas the EU is more cautious, often regulating or banning an additive if there’s even a small chance of it being dangerous.”
At the heart of the differing approaches of FDA and EFSA is the concept of hazard versus risk. The European Union tends to emphasize the potential hazard of an additive, focusing on the possibility that it could cause harm. It often leads to strict regulations or outright bans on certain additives if there is any uncertainty or dispute about their safety.
In contrast, the United States, through the U.S. Food and Drug Administration (FDA), focuses on risk. The FDA assesses the likelihood that a hazard will actually cause harm under specific conditions of use. This means that an additive might be allowed in the U.S. until there is clear evidence that it poses a significant risk to consumers.
The Precautionary Principle in the EU
The EU’s food safety is guided by the precautionary principle, which suggests that if there is any uncertainty about an additive’s safety, it should not be allowed. As a result, this principle allows the EU to reject food additives based on potential hazards, even when scientific evidence is not conclusive.
In contrast, the FDA operates under the authority of the 1938 Federal Food, Drug, and Cosmetic Act, requiring companies to prove that a new additive is safe before it can be used. However, if an additive is recognized as generally safe by experts, companies can use it without FDA approval, provided they submit sufficient evidence. While the U.S. is often seen as more lenient, there are additives permitted in the EU that the FDA does not allow, such as the color additive Green S, made from coal tar.
Implications for Food Additive Manufacturers
These varying regulatory frameworks have practical implications for food manufacturers, especially those operating in multiple markets. Products may need to be reformulated to meet the specific requirements of each region. For example, beverage manufacturers may have to create different formulations for beverages sold in the EU and the U.S. due to differing restrictions on color additives.
While it’s been common practice to create different formulations for different markets, the emergence of state-level regulations in the U.S., such as California’s new food safety law, is a growing concern. The California Food Safety Act, which bans certain additives starting in 2027, mirrors the EU’s precautionary approach. This shift could create challenges for manufacturers, who may need to customize products not just for different countries but also for different states within the U.S. The cost and complexity of customizing formulations state by state can be a huge worry.
Another challenge food manufacturers face is the growing influence of consumer pressure on regulatory decisions. The trend of many of the current bans for food additives driven by consumer fears rather than scientific evidence can be quite problematic as it stifles innovation and makes it harder for companies to develop new products.
How Can Food Additive Manufacturers Prep for this Scenario?
The contrasting approaches to food safety in the U.S. and EU create consequential bottlenecks for food additive manufacturers, particularly those looking to innovate. Navigating these regulatory landscapes requires a deep understanding of both hazard and risk, as well as the ability to adapt to differing consumer attitudes and emerging state-level regulations.
As the industry evolves, finding common ground in food safety standards may be key to ensuring that innovation can continue while protecting public health. Some experts advocate for a more harmonized approach to food safety regulation. Codex Alimentarius, a collection of international food standards, guidelines, and codes of practice, takes a holistic approach to assessing whether an additive is a risk or not and takes a global view that an ingredient is acceptable to use at a certain level and is safe for human consumption, considering the best scientific evidence available. A consolidated global approach could make it easier for food additive manufacturers to develop and market products worldwide while ensuring that safety remains a top priority.
Tech-enabled food safety management solutions are coming forth as valuable tools for food additive manufacturers by streamlining the process of ensuring compliance with specific regulations across different markets and facilitating the management of detailed specifications and supplier information.
Utilizing adequate technologies, manufacturers can more effectively monitor and document the safety of their products, ensuring that all ingredients meet the necessary standards. This not only helps to steer through the intricate web of global food safety strategies but also supports innovation by allowing manufacturers to respond swiftly and accurately to new requirements and market demands.
Smart Food Safe Solutions to Enhance Food Additive Safety for the Global Market
Smart Food Safe‘s digital food safety management solutions provide food additive manufacturers with a comprehensive platform to navigate the varying regulatory landscapes of both the U.S. and EU. By integrating real-time monitoring, simplified compliance management, and consolidated documentation, Smart Food Safe ensures that manufacturers can easily align their products with the differing food safety standards of each region.
Furthermore, Smart Food Safe’s software modules such as Smart Supplier and Smart Spec streamline the management of supplier information and product specifications, enabling food additive manufacturers to maintain detailed, up-to-date records that can be quickly accessed and shared with regulatory bodies. This ensures swift compliance with emerging state-level regulations and helps them remain agile in the face of evolving market demands, reducing time-to-market for new products while safeguarding public health.

